Compensatory damages
A number of states and the federal government use “compensatory mitigation” to replace or enhance wetland habitats that are destroyed or significantly impacted by the construction or widening of highways.
For example, the Maryland State Highway Administration (MSHA) has restored or built over 200 acres of wetlands since 2007. Such mitigation provides many potential environmental benefits, including restoring or enhancing wetlands functions and maintaining existing levels of biodiversity in areas impacted by road construction.
Recently, however, questions have been raised regarding whether such wetlands mitigation projects may have negative ecological consequences as well as benefits. One recent concern is the potential for wetlands mitigation to increase the spread of what are termed “emerging wildlife diseases.”
Jumped into action
Emerging wildlife diseases are fungal, bacterial or viral infectious diseases that negatively impact wildlife populations. They are called “emerging” because their distribution and prevalence is often enhanced by anthropogenic factors, including international trade in wildlife species, habitat alterations from human development and the direct spreading of disease vectors by humans.
A good example of the latter effect is the spread of the White-Nose Syndrome (WNS) in bat populations in the U.S. and Canada. This fungal disease, which has killed millions of bats since 2007, is largely spread via bat-to-bat transmission, but the disease has “jumped” to many distant localities, likely because of human visitation to caves. Without knowing what they are doing, cave explorers visiting a contaminated cave pick up fungal spores on their boots and equipment and can then spread the disease to uncontaminated caves quite distant from the original site. Thus, visitors to caves are asked to decontaminate their equipment and clothing after leaving a site, and many caves in the U.S. have been closed to visitation entirely.
Analysis was performed to determine the prevalance and potential spread of Ranavirus in species at MSHA-constructed wetlands.
Ranavirus ruckus
A less well-known example of a wildlife disease whose spread may be at least partially caused by humans is Ranaviral disease. This disease is caused by a DNA-based virus known as Ranavirus, and is known to cause high levels of mortality in fish, turtles and amphibians, especially frog tadpoles and salamander larvae. Signs of the disease in amphibians include hemorrhaging from the ventral surface (the belly) and erratic swimming.
A possible link between the spread of Ranavirus and wetlands mitigation was found by researchers from Towson University during a recent study examining the environmental impacts of the InterCounty Connector project in Maryland during 2008-2011. As part of the mitigation process, officials from Montgomery County, Md., constructed two small wetlands adjacent to the newly built highway during the fall of 2010. The following spring, Towson University (TU) researchers discovered a large outbreak of Ranaviral disease in both the newly constructed wetlands and in a nearby natural wetlands. Within three days of discovering the outbreak, essentially all of the amphibians in these wetlands were dead from Ranavirus infection, a finding confirmed by analysis from the USGS National Wildlife Health Center in Wisconsin. This level of mortality and rate of onset is not uncommon for Ranavirus outbreaks.
Based on this outbreak and findings from elsewhere in the U.S., researchers hypothesized that if constructed or enhanced wetlands experience higher rates of Ranavirus infection than natural wetlands, then constructed wetlands could potentially be contributing to the spread of Ranavirus across the landscape. Because the virus is apparently carried among sites via animals or, for short periods, on human boots or equipment, increasing the number of wetlands in a given landscape could enhance the probability of the virus being transmitted over longer distances.
To address the question of the role (if any) between constructed wetlands and disease outbreak, the MSHA funded a study by TU and the University of Central Florida (UCF) in 2014-2015. The study was designed to determine (a) whether the prevalence of Ranavirus is associated with wetlands built as part of highway construction projects; (b) whether such rates of prevalence are higher than for natural wetlands; and (c) to examine the relationship between habitat variables and the prevalence of Ranavirus. Combined with additional data from current and future studies, these data can eventually be used in a best practices set of recommendations to the MSHA to minimize the odds that constructed or restored wetlands are contaminated with the disease.
TU staff reviewed the available MSHA database of constructed wetlands and compiled a catalogue of potential field sites. On-site inspection of 60 suitable sites began in February 2014. This was the maximum number of potentially suitable sites available within a logistically feasible geographic area. Site inspections consisted of an assessment of suitability of the sites using specific criteria, including presence/absence of amphibian breeding, proximity to roads, proximity to natural habitats for amphibian dispersal, water quality, and a preliminary assessment of wetland depth and likely hydroperiod.
TU staff made repeated sampling trips to the selected wetlands starting in mid-February 2014 and 2015, with visits continuing until July 2015. The wetlands were sampled at the onset of the breeding season for Spotted Salamander and Wood Frog egg masses and tadpoles to document the presence of active amphibian use. Return visits to these sites were conducted starting about 60 days after initial identification to determine the degree of tadpole development. More frequent visits (normally two times per week) were used to determine whether there was a Ranavirus-caused mortality event, which is characterized by easily observed signs of disease such as the aforementioned hemorrhaging from the ventral surface and abnormal swimming behavior.
Because other diseases may cause sudden morality events in the field, it was essential the causative agent be identified correctly. This can only be done via DNA analysis using Polymerase Chain Reaction (PCR) to directly test for the presence of Ranavirus DNA in an animal sample. The research team provided selected samples of tadpoles possibly infected with Ranavirus to the research laboratory at UCF for PCR testing to confirm the presence of the virus.
Rising rates
This study showed that infection rates in MSHA-constructed wetlands in Maryland were among the highest reported occurrences in the region, with 54% of study ponds having either Ranavirus detection via PCR analysis or an observed outbreak event. In comparison, non-SHA wetland sites in the state had a much lower infection rate (27%). Delaware and New Jersey also had high detection rates, with Ranavirus being found in 57% of sites in Delaware and 48% in New Jersey. Pennsylvania and Virginia fared much better, with both states having less than 5% of sites testing positive for the virus.
However, interpreting these apparent differences must be done cautiously. TU personnel sampled most wetlands twice per week, which was more frequently than many sites included in the regional samples. This means that the rate of detection of die-offs was more likely at SHA sites in Maryland than in the regional samples due to the rapid onset of an outbreak and rate of decomposition of the amphibian larvae. In some cases, the entire process from detection to complete mortality may only last three days. In light of this, results should be viewed as a conservative estimate of Ranavirus prevalence, with a key takeaway message from the study being the necessity of frequent monitoring.
With this information in mind, it is difficult to draw firm conclusions. However, the data do suggest that SHA-constructed wetlands have a higher rate of Ranavirus infection compared to non-SHA-constructed wetlands. It is the belief of the authors that additional work will show the pathogen to be more widespread than currently documented, which would likely decrease the difference in Ranavirus presence between states.
Additionally, quantitative PCR analysis of viral DNA unexpectedly found the concentrations of virus (“infection intensity”) in specimens collected from SHA sites to be extremely high. In the SHA wetlands where samples of dying tadpoles were collected and used for DNA analysis, infection intensities ranged from the tens of millions to the billions of copies of the virus. These levels are several orders of magnitude higher than those seen in some other recent studies. No explanation for these differences or the potential impact to amphibian communities is immediately apparent, but one possibility is that the viral strains found in these wetlands are especially virulent, replicating unhindered and leading to especially rapid outbreaks.
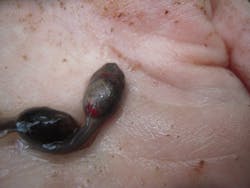
A tadpole carrying Ranavirus. Such specimens were evaluated for their propensity to spread disease.
Drying out
A key component to understanding a disease is to identify factors allowing the pathogen to spread and those factors that trigger an actual outbreak. To test this, several models were used to evaluate what elements could be linked to Ranavirus including environmental and biological variables.
Statistical analysis showed that models using the likelihood of wetlands drying and distance from the nearest stream could be important for determining the occurrence of Ranavirus infections. However, these models could not be statistically differentiated compared to a “null” model. Other variables such as wetland age, depth, temperature, size or distance from roads did not appear to influence the rate of occurrence of Ranavirus.
These links are statistically weak, but biologically compelling. Ponds that dry out at least once a year are approximately 30% more likely to have an outbreak than those that do not. Additionally, the further a wetland is located from a stream, the less likely it is to have Ranavirus. The exact reasons as to why this may be the case are not certain at this time; however, some hypotheses have been suggested.
The link between wetlands that dry out once per year (i.e., vernal pools, ephemeral wetlands) and an increased risk of Ranavirus is at first puzzling, as the virus cannot survive under dry conditions. The answer may reside in the species that frequent these ponds. Many amphibians in this region depend on these temporary habitats to breed, specifically Wood Frogs and Spotted Salamanders. These two species also are especially sensitive to Ranavirus, and could be serving as optimal hosts for the virus, thus magnifying the amount of viral concentrations within these systems. However, this scenario would require a wetland to be recolonized by the virus each year, as it should die off annually as the pond dries.
Distance from streams may indicate geographic isolation from a potential source of infection, and could link into the scenario outlined above. If streams are functioning as a Ranavirus source, then species carrying the disease would be less likely to travel hundreds or thousands of meters to these more distant wetlands. Attention has been given to Green Frogs, which have shown to be resistant to the virus, and also are known to travel large distances to find suitable foraging and breeding grounds.
Again, these correlations are preliminary and additional research will need to be done to confirm these hypotheses. High variability of environmental and biotic factors among years adds to this complexity, such as variation in species presence, rate of larval development, temperature and rain accumulation. Furthermore, the presence of the virus does not guarantee an outbreak in a given year, and an outbreak in one year does not necessarily mean an outbreak will occur the following year. Robust conclusions are difficult to claim based on the current two years of data.
Overall, these data suggest a possible link between wetland hydroperiod and distance to streams to the presence of Ranavirus, and hint that SHA-constructed wetlands could have a higher risk of Ranavirus presence compared to non-SHA-constructed wetlands. However, a larger sample size of ponds would be needed to provide a more robust test of this association.
Our understanding of this disease is still in the early stages of development, and several fundamental questions relating to infection vector, distribution and development of resistance in populations still require investigation. Studies of Ranavirus in other states further support the hypothesis that Ranavirus has the potential to affect amphibian and reptile populations in serious ways. A better understanding of how this disease spreads as a function of wetlands mitigation projects represents an urgent conservation and management need.
Make a clean break
The researchers advocate that construction staff take care to minimize the risk of transporting this disease among wetlands. A key recommendation is to decontaminate equipment and boots with either a 3% bleach solution or a 1% Novalsan solution every time staff move between wetland sites. Keeping a sprayer with a premixed solution on days when visiting multiple sites is a quick way to minimize risk. Construction staff also should keep an eye out for possible disease outbreaks. If you believe you are seeing an outbreak, contact your state wildlife agency.
-----------------
Seigel is a professor of biology at Towson University. Saffell is an environmental scientist at Environmental Systems Analysis Inc. Nein is an environmental scientist at JMT Inc., Durkin is a graduate student in biology at Towson University. Martin is a Ph.D. student in evolution, ecology, and organismal biology at The Ohio State University. Lawrance is a biological lab manager at the University of Central Florida. Savage is an assistant professor of biology at the University of Central Florida.